NEWS AND INFORMATION FROM SYSTEC & SOLUTIONS GMBH
All news on cleanroom hardware, branch events, and Systec & Solutions at a glance.
Here you can find the latest information on our products and the company and read all about forthcoming and past events.
- 05.02.2026
Whitepaper: Hygienic design and microbiological safety of HMIs for cleanroom use
Concise insights into technical and regulatory requirements for cleanroom and life science applications, presented in a clear and practical manner.
- 21.07.2025
Summer party 2025: a memorable day for employees, families and friends
This year's Systec & Solutions GmbH summer party in Karlsruhe took place in glorious sunshine, a great event that brought together colleagues, their families and friends.
- 07.07.2025
Ralf Kreuzer - New Head of Business Development APAC
We are delighted to introduce to you Ralf Kreuzer, who recently joined us as Head of Business Development APAC. He brings valuable experience that will strongly support our continued growth across the Asia-Pacific region.
- 02.07.2025
Success Story: Excella optimizes cleanroom production with modern HMI technology
How can cleanroom processes be made safer, more efficient and fully digitalized? A project with our customer Excella shows it!
- 04.04.2025
Systec & Solutions Showcases Cleanroom Solutions at INTERPHEX in New York
Systec & Solutions looks back on a highly successful debut at INTERPHEX 2025. The renowned trade show for the pharmaceutical and biotechnology industries took place from April 1–3, 2025, at the Javits Center in New York City.
- 19.03.2025
The Systec & Solutions Group expands to the United States
The Systec & Solutions Group, a leading provider of high-quality hardware and software solutions for cleanroom environments, is excited to announce the opening of its new U.S. site in Apex, North Carolina.
- 24.11.2024
Life Science Ready Days 2024
Have you missed it? Experience the highlights of our LSR Days 2024 with exciting presentations, workshops and networking. Let yourself be inspired!
- 09.10.2024
The new PILOT - a leap forward in design and functionality
In a new, slim design, with improved functionality and groundbreaking innovations for your life science requirements: Discover the PILOT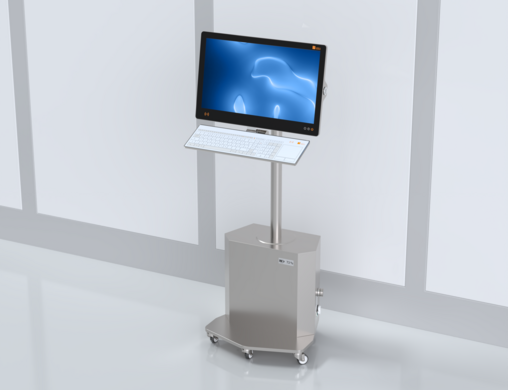
- 20.08.2024
PACK EXPO 2024

- 11.06.2024
Achema 2024
Discover our innovative HMI systems in a virtual cleanroom environment with VR technology?![[Translate to English:] Systec Produkte im virtuellen Raum als 3D Modell der Firma Pharmaplan](/fileadmin/_processed_/5/8/csm_systec-produkte_3D-raum_pharmaplan_e3e093774e.png)
- 23.05.2024
Optimizing tilt angle for HMI and display ergonomics: a science-based approach
Unveiling the impact of tilt angles on human-machine interfaces (HMIs) and displays - 14.05.2024
CONNECT BOX for TABLET with Surface Pro 8 and RFID/NFC reader
User identification is now even easier and faster on the TABLET with the new CONNECT BOX for Surface Pro 8 with integrated RFID/NFC reader.
- 02.05.2024
TROLLEY SLIM with TABLET DOCKING HINGE and power connection
Our innovative mobile workstation, the TROLLEY SLIM, redefines mobility in cleanrooms.
- 06.02.2024
Dr. Martin Printz becomes new Managing Director at Systec & Solutions
Systec & Solutions is pleased to announce the appointment of Dr. Martin Printz as additional Managing Director as of February 1, 2024.
- 06.02.2024
Martin Storm Marum becomes Managing Director of Systec & Solutions ApS
Systec & Solutions ApS is pleased to announce that Martin Storm Marum will be the new Managing Director as of February 1, 2024.
- 15.08.2023
Visit us at LOUNGES 2022 in Karlsruhe
It's finally time! Trade fairs such as the LOUNGES CLEANROOM PROCESSES in Karlsruhe are once again taking place.
- 15.08.2023
HMI systems
for cleanroom environmentsWe now offer the WAVE®, PILOT®, and CONTROL product ranges in the pre-configured versions LIGHT, ENDPOINT, ALLROUND, and PERFORMANCE.
- 15.08.2023
In its third generation – the new HMI CONTROL for wall mounting in the cleanroom
At a high level of technical performance, the HMI has been upgraded, now featuring the EASY CLICK MAGNETIC MOUNT mounting technology as standard. By using the magnetic ...
- 15.08.2023
UL certification for selected HMI systems from the WAVE and PILOT model ranges
Selected HMIs from the WAVE and PILOT model ranges (17.3" to 24" versions) are now UL-certified and will carry the UL-certified logo.